
Consumers naturally assume that the products they purchase and use daily are safe, a fundamental expectation built on trust and regulatory oversight. We rely on rigorous safety measures and diligent manufacturing processes to prevent defective items from reaching market shelves, ensuring our well-being and peace of mind. However, history repeatedly shows that this trust can be broken, often with devastating consequences that leave lasting scars on public confidence and industry practices.
Despite advances in product safety, the reality is that faulty products can and do impact millions. For instance, in 2023 alone, the National Electronic Injury Surveillance System (NEISS) reported over 12.9 million injuries associated with consumer products in the U.S., spanning categories from toys to automobiles and household appliances. Such statistics underscore a persistent challenge in safeguarding public health against product-related hazards, demonstrating that vigilance remains crucial in our consumption habits.
Often, the true scope of injuries and dangers linked to defective products only becomes apparent after widespread harm has occurred, prompting a formal recall either voluntarily by companies or under mandate from institutions like the U.S. Food and Drug Administration. These recalls, while crucial for public safety, represent moments when corporate responsibility faltered, revealing vulnerabilities in the systems designed to protect us. Ahead, we delve into some of the most notorious product safety scandals in consumer history, aiming to understand the profound impacts these events had on lives, industries, and the evolution of consumer protection in America.

1. **Takata Air Bag Recall (2008 and counting)**
The ongoing saga of faulty airbags manufactured by Takata stands as arguably the biggest and most notorious product recall in history, a crisis that has tragically claimed lives and injured hundreds globally. What began quietly almost a decade ago has since ballooned into a global catastrophe, involving virtually every major automaker on the planet. The core issue lies with defective airbag inflators that can explode unexpectedly, ejecting dangerous, shrapnel-like material upon deployment, turning a safety device into a deadly projectile.
The sheer scale of this recall is staggering. The National Highway Safety Transportation Board (NHTSA) anticipates that the recall will ultimately encompass over 37 million vehicles in the U.S. alone, involving a staggering 49.5 million inflators. Globally, the numbers are even more immense, with 100 million inflators under recall. Regulators have projected that it could take until 2023 to identify and repair every single vehicle equipped with a faulty Takata airbag, underscoring the immense logistical challenge and the prolonged risk to consumers.
The human cost has been devastating, with these defective airbags linked to at least 20 deaths and 200 injuries as of 2017, according to the NHTSA. Financially, the impact has been equally profound; Takata estimated total recall costs at $24 billion in 2016, a figure that remains subject to change given the ongoing nature of the issue. The Japanese company ultimately filed for bankruptcy in 2017, a stark indicator of the catastrophic burden this recall placed on its operations and existence.
This scandal serves as a sobering reminder of how a single component supplier’s failure can reverberate across an entire industry, endangering millions of consumers worldwide. It underscores the critical need for robust quality control, transparent reporting, and diligent oversight throughout complex global supply chains. The Takata recall has undeniably reshaped how automakers and regulators approach component sourcing and safety compliance, leaving an indelible mark on automotive safety standards and the public’s perception of vehicle reliability.

2. **Volkswagen’s Diesel Engine Scandal (2015)**
The automotive world was collectively stunned in 2015 when German car giant Volkswagen, a brand long associated with precision engineering and reliability, was caught in a massive scandal dubbed “Dieselgate.” This deeply troubling episode revealed that the company had deliberately cheated on diesel emissions tests, a deception that not only violated environmental regulations but also profoundly betrayed consumer trust and ethical conduct.
The core of the scandal involved sophisticated software, or “defeat devices,” installed in millions of Volkswagen’s turbocharged diesel engines. This software was designed to detect when a vehicle was undergoing an official emissions test, at which point it would activate full emissions controls to meet regulatory standards. However, under real-world driving conditions, these engines would revert to a different operating mode, emitting pollutants—specifically nitrogen oxide (NOx)—at levels up to 40 times higher than those permitted under U.S. standards, directly impacting air quality and public health.
The fallout from this deliberate deception was immense and immediate. Volkswagen was compelled to recall 11 million vehicles globally, a logistical and financial nightmare. Beyond the recalls, the company was forced to set aside more than $18 billion to cover a wide array of costs, including recall expenses, legal claims, and other related penalties. This included a substantial $14.7 billion settlement to compensate car owners and address environmental harm in the U.S. alone. Additionally, Volkswagen agreed to pay $2.7 billion to fund efforts aimed at reducing NOx emissions in severely smog-affected areas and pledged to invest $2 billion in the manufacturing and promotion of electric vehicles.
The scandal not only led to a significant plunge in Volkswagen’s stock prices—by a third initially, taking two years to recover—but also prompted the resignation of its CEO and severely tarnished the brand’s reputation for honesty and integrity. It sparked critical debates about corporate ethics, the enforceability of environmental regulations, and the accountability of major manufacturers, forcing a global reevaluation of emissions testing protocols and strengthening regulatory resolve against corporate malfeasance.
Read more about: Beyond the Showroom Shine: 14 Automotive Design Fails That Rocked the Industry

3. **Merck’s Vioxx Recall (2004)**
In the pharmaceutical industry, the recall of Merck’s arthritis painkiller, Vioxx, stands as one of the deadliest and most financially staggering product safety scandals in history. When Vioxx was first introduced to the market in 1999, it was hailed as a revolutionary breakthrough medication, quickly becoming a blockbuster drug with widespread use among arthritis sufferers. The initial promise of effective pain relief, however, obscured a darker reality that would soon emerge with devastating consequences for public health.
Five years after its launch, in September 2004, Merck made the difficult decision to voluntarily pull Vioxx from the market. This drastic action came after studies revealed a deeply alarming association: Vioxx greatly increased the risk of fatal heart attacks and strokes. By the time of the recall, an estimated 20 million Americans had already taken the drug, leaving a vast number of individuals potentially exposed to its severe side effects. Later research would tragically estimate that up to 140,000 Americans experienced heart attacks linked to Vioxx, resulting in a staggering 88,000 deaths.
The financial repercussions for Merck were equally immense. The pharmaceutical giant faced a torrent of lawsuits and settlements, with costs skyrocketing into the billions. In 2007, Merck settled a class-action lawsuit for a colossal $4.85 billion. This was followed by a $950 million settlement with the Department of Justice in 2011 to resolve civil and criminal allegations related to the drug’s marketing. By 2016, shareholders also settled a class-action lawsuit for $830 million. Statista estimated that, when all other expenses were factored in, the Vioxx recall cost the company an astonishing $8.9 billion.
The news of the recall sent shockwaves through the financial markets, with shares in Merck initially tumbling by 27% and languishing for two years before recovering. Beyond the financial impact, the Vioxx scandal profoundly shook public confidence in pharmaceutical safety and regulatory oversight. It prompted intense scrutiny of drug testing protocols, post-market surveillance, and the process by which new medications are approved and monitored for long-term safety, demanding greater vigilance from both pharmaceutical companies and regulatory bodies like the FDA.

4. **Firestone Tires and Ford (2000)**
The early 2000s saw a devastating product safety crisis unfold between two automotive giants: Bridgestone’s Firestone Tire and Rubber Company and Ford Motor Company. This bitter corporate feud stemmed from allegedly defective tires installed predominantly on Ford SUVs and pickup trucks, particularly the Ford Explorer, and resulted in a tragic loss of life and countless injuries across the U.S. The crisis brought into sharp focus the critical importance of component reliability in complex products like automobiles.
The core of the problem lay in tread separation issues with Firestone Wilderness AT tires, which were standard equipment on many Ford Explorer models. These separations, occurring at highway speeds, often led to catastrophic blowouts, causing vehicles to lose control and roll over. The National Highway Traffic Safety Administration (NHTSA) reported that more than 100 deaths and hundreds of injuries involving the Ford Explorer were directly attributed to these tire failures. Overall, the crisis was linked to 271 deaths and more than 800 injuries in the U.S. alone, painting a grim picture of the human toll.
As the crisis escalated, a highly public and acrimonious blame game ensued between Firestone and Ford. Firestone contended that Ford’s vehicle design, particularly the Explorer’s tendency to roll over, exacerbated the problem. Ford, in turn, maintained that the Firestone tires were inherently defective. Ultimately, both companies found themselves “on the hook” for the catastrophic failures. Firestone was forced to recall 6.5 million tires, while Ford took the extraordinary step of recalling and replacing an additional 13 million Firestone tires on its vehicles.
The financial consequences for both companies were immense. The tire recall and the ensuing corporate fallout cost Firestone’s parent company, Bridgestone, an estimated $2 billion. Ford informed its shareholders in 2001 that its recall of 13 million tires would cost the company approximately $3 billion. Additionally, Ford faced substantial legal liabilities, including $600 million in lawsuits from victims and their families. The Firestone brand, though it survived, saw its reputation severely damaged, and its century-long relationship with Ford was irrevocably severed. This scandal profoundly impacted consumer perception of vehicle safety, particularly for SUVs, and led to significant regulatory reforms, including the TREAD Act.
Car Model Information: 2022 Ford Explorer Platinum
Name: Ford Explorer
Caption: Sixth-generation Ford Explorer
Manufacturer: Ford Motor Company
Production: 1990–present
ModelYears: 1991–present
Class: unbulleted list
Chassis: unbulleted list
Predecessor: Ford Bronco II
Successor: Ford Territory (Australia)
Categories: 2000s cars, 2010s cars, 2020s cars, All-wheel-drive vehicles, All Wikipedia articles in need of updating
Summary: The Ford Explorer is a range of SUVs manufactured by the Ford Motor Company since the 1991 model year. The first five-door SUV produced by Ford, the Explorer, was introduced as a replacement for the three-door Bronco II. As with the Ford Ranger, the model line derives its name from a trim package previously offered on Ford F-Series pickup trucks. As of 2020, the Explorer became the best-selling SUV in the American market.
Currently in its sixth generation, the Explorer has featured a five-door wagon body style since its 1991 introduction. During the first two generations, the model line included a three-door wagon (directly replacing the Bronco II). The Ford Explorer Sport Trac is a crew-cab mid-size pickup derived from the second-generation Explorer. The fifth and sixth generations of the Explorer have been produced as the Ford Police Interceptor Utility (replacing both the Ford Crown Victoria Police Interceptor and the Ford Police Interceptor Sedan).
The Explorer is slotted between the Ford Edge and Ford Expedition within North America’s current Ford SUV range. The model line has undergone rebadging several times, with Mazda, Mercury, and Lincoln each selling derivative variants. Currently, Lincoln markets a luxury version of the Explorer as the Lincoln Aviator.
For the North American market, the first four generations of the Explorer were produced by Ford at its Louisville Assembly Plant (Louisville, Kentucky) and its now-closed St. Louis Assembly Plant (Hazelwood, Missouri). Ford currently assembles the Explorer alongside the Lincoln Aviator and the Police Interceptor Utility at its Chicago Assembly Plant (Chicago, Illinois).
Get more information about: Ford Explorer
Buying a high-performing used car >>>
Brand: Ford Model: Explorer
Price: $33,399 Mileage: 35,792 mi.

5. **Johnson & Johnson’s Tylenol Recall (1982)**
Widely recognized as “the recall that started them all,” the 1982 Tylenol poisoning incident remains a seminal event in product safety history, fundamentally reshaping how corporations manage crises and how consumer products are packaged. This tragic case of product tampering unfolded in the Chicago area, where seven people died after ingesting Extra-Strength Tylenol capsules that had been maliciously laced with cyanide. The incident sent shockwaves across the nation, exposing a terrifying vulnerability in everyday consumer products.
The immediate aftermath was one of widespread panic and fear, as consumers grappled with the terrifying realization that everyday medications could be deadly. The perpetrator of the tampering was never found, leaving a chilling unsolved mystery at the heart of the crisis. Johnson & Johnson (J&J), the manufacturer of Tylenol, faced an unprecedented challenge to its brand and public trust. At the time, Tylenol was J&J’s best-selling product, accounting for 17% of the company’s profits, making the stakes incredibly high for the corporation’s future.
J&J’s response to the crisis has since become a benchmark for exemplary corporate crisis management. The company took swift and decisive action, spending more than $100 million to recall 31 million bottles of Extra-Strength Tylenol nationwide. This voluntary recall was an extraordinary measure for its time, demonstrating a clear prioritization of consumer safety over immediate financial concerns. Adjusted for inflation, this recall cost roughly $250 million, a substantial sum, yet it was crucial for regaining public confidence.
Beyond the recall, J&J implemented groundbreaking changes that would set new industry standards. They introduced tamper-evident packaging for all over-the-counter medications, a design innovation that soon became a federal law, requiring all similar products to adopt comparable safeguards. This proactive measure not only helped restore consumer confidence in Tylenol, which recovered its market share within two months despite an initial stock price swoon, but also ushered in a new era of product security across various industries. This incident permanently altered consumer expectations for product safety and packaging.

6. **The Ford Pinto Fuel Tank Scandal (1970s/1978)**
The Ford Pinto fuel tank scandal remains one of the most notorious and frequently cited cautionary tales in business ethics, fundamentally transforming auto safety standards and serving as a stark reminder of the dangers of prioritizing profits over human lives. During the years 1971 to 1980, Ford sold over 3 million Pinto model vehicles, a popular compact car of its era. However, the design harbored a critical and deadly flaw that would lead to tragic consequences.
The issue centered on the Pinto’s defective fuel tank, which was placed too close to the rear axle, making it highly susceptible to rupture and catastrophic fires in even low-speed rear-end collisions. Disturbingly, internal documents later revealed that Ford was aware of this design flaw before the car was released. A notorious internal cost-benefit analysis, famously known as the “Pinto memo,” indicated that the cost of redesigning and fixing the fuel tank (estimated at $11 per vehicle) outweighed the potential lawsuit payments for deaths and injuries. The company, at the time, made the shocking decision that it was “better to pay for injury claims than to fix the issue.”
This corporate calculation had horrific real-world consequences. It is estimated that around 180 deaths can be attributed to Pinto’s fuel tank fires, alongside numerous devastating injuries. While the recall of 3.1 million Pinto Ford vehicles officially occurred in 1978, years after the company first became aware of the defect, the damage to Ford’s reputation and consumer trust was profound. One highly publicized case involved a man whose Pinto burst into flames during a low-speed accident, leading to a jury awarding $125 million in damages, though this was later reduced.
The scandal ignited a firestorm of public outrage and legal challenges, placing a harsh spotlight on corporate accountability and product liability. It became a powerful catalyst for increased scrutiny of automotive safety and led to significant reforms in vehicle design standards. The case highlighted the critical importance of ethical decision-making throughout the product development process, emphasizing that a company’s financial interests should never supersede its fundamental responsibility to ensure the safety of its customers. The Ford Pinto scandal continues to be a cornerstone of business ethics courses, illustrating the severe repercussions when a company consciously chooses potential lawsuit payments over implementing necessary safety fixes.
Car Model Information: 1980 Ford Pinto WAGON
Name: Ford Pinto
Caption: Ford Pinto
Manufacturer: Ford Motor Company
Aka: Mercury Bobcat
Production: September 1970 – July 1980
ModelYears: 1971–1980 (Pinto),1974–1980 (Bobcat)
Assembly: Edison, New Jersey,Milpitas, California
Designer: Robert Eidschun (1968)
Class: Subcompact car
BodyStyle: Sedan (automobile),sedan delivery,station wagon,hatchback
Related: #Mercury Bobcat (1974–1980),Ford Mustang (second generation)
Layout: Front-engine, rear-wheel-drive layout
Chassis: Unibody
Engine: unbulleted list
Abbr: on
Disp: Ford Cologne engine
Transmission: unbulleted list
Wheelbase: 94.0 in
Length: 163 in
Width: 69.4 in
Height: 50 in
Weight: convert
Predecessor: Ford Cortina#Mark II (1966–1970)
Successor: Ford Escort (North America)
Categories: 1980s cars, Articles with short description, Cars discontinued in 1980, Cars introduced in 1970, Commons category link from Wikidata
Summary: The Ford Pinto is a subcompact car that was manufactured and marketed by Ford Motor Company in North America from 1970 until 1980. The Pinto was the first subcompact vehicle produced by Ford in North America.
The Pinto was marketed in three body styles throughout its production: a two-door fastback sedan with a trunk, a three-door hatchback, and a two-door station wagon. Mercury offered rebadged versions of the Pinto as the Mercury Bobcat from 1975 until 1980 (1974–1980 in Canada). Over three million Pintos were produced over its ten-year production run, outproducing the combined totals of its domestic rivals, the Chevrolet Vega and the AMC Gremlin. The Pinto and Mercury Bobcat were produced at Edison Assembly in Edison, New Jersey, St. Thomas Assembly in Southwold, Ontario, and San Jose Assembly in Milpitas, California.
Since the 1970s, the safety reputation of the Pinto has generated controversy. Its fuel-tank design attracted both media and government scrutiny after several deadly fires occurred when the tanks ruptured in rear-end collisions. A subsequent analysis of the overall safety of the Pinto suggested it was comparable to other 1970s subcompact cars. The safety issues surrounding the Pinto and the subsequent response by Ford have been cited widely as business ethics and tort reform case studies.
Get more information about: Ford Pinto
Buying a high-performing used car >>>
Brand: Ford Model: Pinto
Price: $5,951 Mileage: 107,000 mi.

7. **General Motors’ Ignition Switch Recall (2014)**
The year 2014 cast a dark shadow over General Motors, as the automotive giant confronted a monumental product safety crisis. Faulty ignition switches, a seemingly minor component, possessed the alarming capability to shut down a vehicle’s engine without warning. This action disabled critical safety systems like power steering, brakes, and airbags, transforming a safety feature into a profound hazard for drivers and passengers.
The human cost was devastating. These defective switches were directly linked to at least 124 deaths and over 248 injuries. Compounding the tragedy, investigations revealed GM had been aware of these safety issues as early as 2004, a full decade before widespread recalls. This delayed response highlighted a severe lapse in corporate responsibility, eroding public trust in a foundational American brand.
Financially, the repercussions for General Motors were immense, reflecting the scale of the recall and subsequent legal liabilities. The company reported recall costs of $4.1 billion. This substantial sum covered $2.8 billion for vehicle repairs, $870 million to settle death and injury claims, and $900 million in a settlement with the Department of Justice for concealing the defect. An additional $874 million charge was taken for future recall costs.
Beyond direct expenditures, GM’s stock price plummeted by about 15% in 2014, underperforming a generally rising market. Long-term investor confidence also suffered. This scandal underscored the critical need for transparent reporting and swift action when safety defects are identified, reshaping industry standards and regulatory expectations.

8. **Toyota’s Floor Mat Recall (2010)**
At the start of a new decade, Toyota, a brand long associated with reliability, faced one of the costliest recalls in automotive history. The crisis originated from a critical design flaw: accelerator pedals could become entrapped or stuck by ill-fitting floor mats, leading to dangerous unintended acceleration. This defect created terrifying scenarios, undermining consumer confidence in Toyota’s celebrated engineering.
The recall encompassed 8.1 million vehicles globally, a logistical challenge of immense proportions. Government investigations disturbingly linked unintended acceleration in Toyota vehicles to the deaths of 89 people over the preceding decade. This grim statistic highlighted the severity of the flaw, transforming a seemingly innocuous component interaction into a fatal hazard across the automotive industry.
Initially, Toyota estimated direct recall costs at $2 billion in 2010. However, the financial fallout dramatically increased four years later. The company agreed to pay a substantial $1.2 billion fine to the U.S. Justice Department to avoid prosecution for allegedly covering up its knowledge of the ill-fitting floor mats and other safety problems. This settlement pointed to concerns regarding corporate transparency and accountability.
The impact on Toyota’s market standing was significant. While shares recovered somewhat, gaining about 60% since late 2009, this lagged far behind the broader S&P 500-stock index, which surged by 155% over the same period. The “floor mat recall” undeniably tarnished Toyota’s reputation for safety, prompting a thorough reevaluation of quality control and reinforcing the need for meticulous design and testing.
Read more about: Beyond the Blink: How a Ford Camera Glitch Uncovers Systemic Engineering Failures and the Lingering Shadows of Automotive Design Mistakes

9. **Pfizer’s Bextra Recall (2005)**
In 2005, pharmaceutical giant Pfizer suffered a major blow with the forced recall of Bextra, an arthritis painkiller. Initially, this COX-2 inhibitor was a best-selling product, generating $1.3 billion in annual sales by 2004. However, its success was abruptly halted when the U.S. Food and Drug Administration (FDA) mandated its withdrawal due to serious concerns over potential heart risks and “life-threatening” skin reactions.
The FDA’s decisive action stemmed from emerging scientific evidence linking Bextra to an increased risk of cardiovascular events, mirroring issues with similar drugs. The presence of severe, potentially fatal adverse reactions indicated that the drug’s benefits no longer outweighed its documented risks. This move underscored the critical role of post-market drug surveillance and regulatory bodies in protecting public health against medications with dangerous side effects.
The recall was just one part of Pfizer’s challenges. In 2009, the company settled civil and criminal allegations of illegally marketing Bextra. The resulting $2.3 billion payout marked the largest healthcare fraud settlement and criminal fine of its kind at the time, exposing aggressive and unethical promotional strategies within the industry.
When accounting for lost sales, fines, settlements, and other costs, the Bextra recall cost Pfizer at least $3.3 billion, according to Statista. Long-term investors who bought shares after the 2009 settlement saw them trail the S&P 500 by approximately 50 percentage points. This scandal highlighted the immense financial and reputational damage resulting from safety concerns and regulatory scrutiny, emphasizing ethical drug development and transparent clinical data.

10. **Samsung’s Galaxy Note 7 Recall (2016)**
In 2016, Samsung, the leading smartphone manufacturer, faced a global product safety crisis with the recall of its flagship Galaxy Note 7. This premium device, intended to be a market leader, was quickly marred by widespread reports of units overheating, catching fire, and even exploding. This unprecedented issue not only endangered consumers but also severely impacted Samsung’s brand and financial standing.
The root cause was defective lithium-ion batteries prone to overheating. Within two months of its August 2016 launch, the U.S. Consumer Products Safety Commission (CPSC) received 96 reports of overheating and fires. These incidents, causing property damage and minor injuries, garnered global headlines. The danger became so pervasive that airlines warned passengers to power off and not charge Note 7s during flights, underscoring the severity of the safety threat.
Samsung responded with two massive recalls. An initial attempt to replace faulty devices failed when replacement units exhibited the same dangerous defect. Samsung then made the extraordinary decision to permanently discontinue the Galaxy Note 7 and recall all 2.5 million units sold globally. This decisive action, taken mere months after launch, reflected the gravity of the battery flaw and the company’s ultimate prioritization of consumer safety.
The financial toll on Samsung was substantial, estimated at $5.3 billion, covering recall costs, lost sales, and discontinuing the product line. Despite this blow, Samsung, a diversified electronics giant, showed remarkable resilience. The company swiftly unveiled the Galaxy Note 8 the following year to strong reviews and robust sales, demonstrating its capacity to recover from a colossal product failure through decisive action and continued innovation in a fast-paced technology market.
Read more about: Navigating the Fallout: 14 Notorious PR Disasters That Tested Brands and Trust
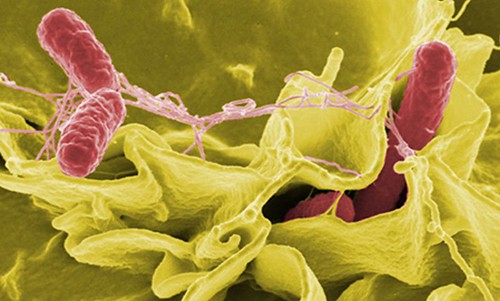
11. **Peanut Corporation of America Salmonella Outbreak (2009)**
A decade ago, the obscure Peanut Corporation of America (PCA) became the epicenter of a massive salmonella outbreak that reshaped U.S. food safety. This devastating crisis claimed nine lives and sickened hundreds, with over 700 confirmed illnesses across multiple states. It triggered one of the largest food recalls in U.S. history, demonstrating how critical lapses in quality control can have widespread, deadly repercussions.
The investigation uncovered appalling conditions at PCA’s processing plants, revealing a shocking disregard for food safety. Evidence emerged of leaky roofs, rampant rodent infestations, and corporate executives knowingly shipping contaminated products despite failed internal safety tests. This deliberate criminal negligence, prioritizing profit over public health, magnified the scandal and intensified outrage, exposing a profound ethical failure.
The fallout extended broadly. Contaminated peanuts and peanut butter were ingredients in over 3,913 different products from roughly 361 companies, prompting an unprecedented wave of recalls. While major brands were unaffected, widespread fear caused consumers to shun peanut butter, driving industry-wide sales down 25%. This collective distrust highlighted the fragility of consumer confidence in food safety.
Legal and financial consequences for PCA were severe. The company declared bankruptcy and ceased operations. Executives faced criminal charges, with a former top executive sentenced to 28 years in prison – the harshest penalty ever for a food safety case. The American peanut industry incurred estimated losses of $1 billion. This scandal was a powerful catalyst for the 2011 Food Safety Modernization Act, shifting regulatory focus to proactive prevention.
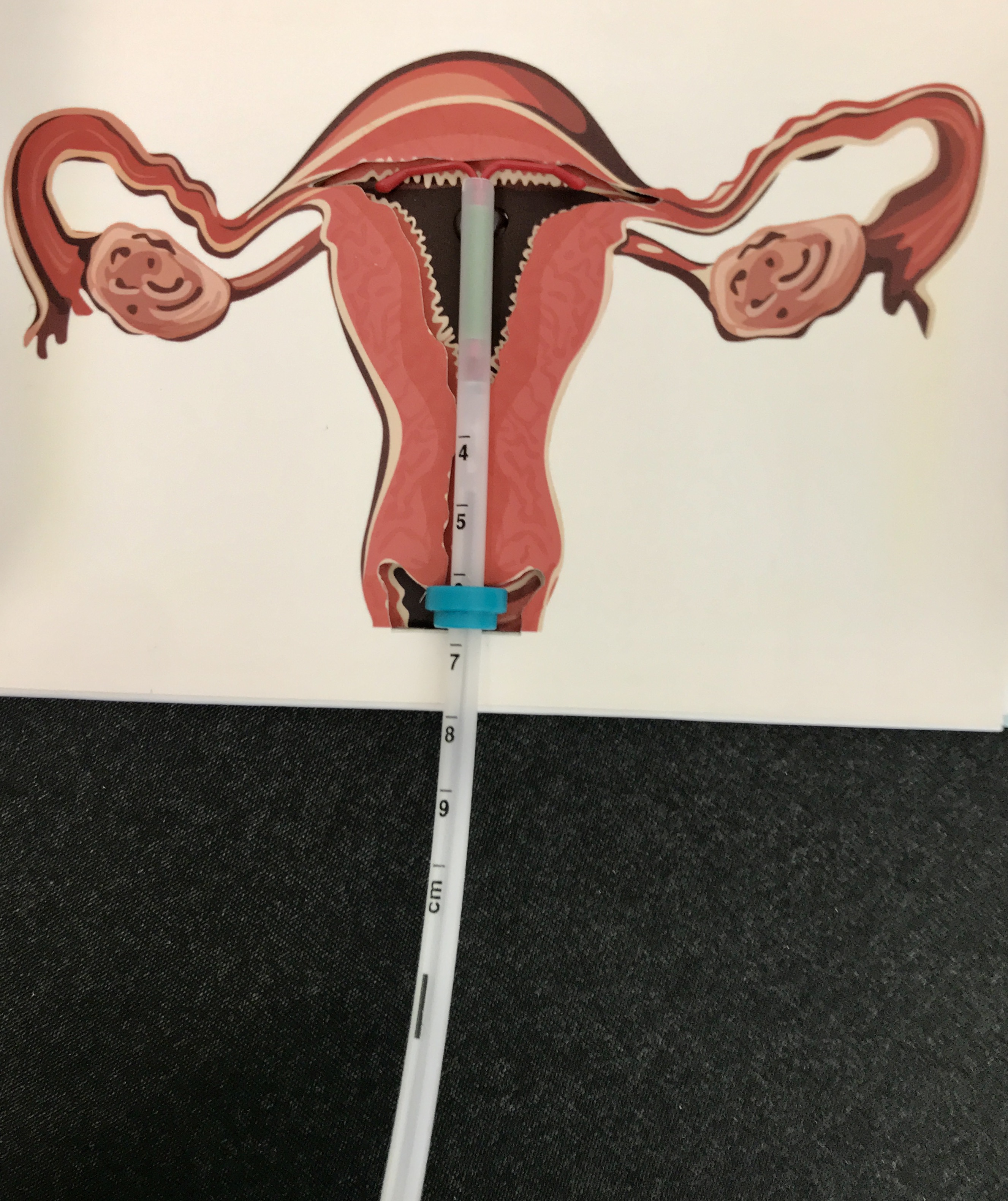
12. **The Dalkon Shield IUD Scandal (1970s)**
In the 1970s, the Dalkon Shield intrauterine device (IUD) created a profound public health crisis for American women, leaving a lasting mark on medical device regulation. Marketed as a safe and effective birth control, this IUD, manufactured by A.H. Robins Company, was soon linked to a devastating array of complications, revealing a critical design flaw that would impact millions.
The Dalkon Shield’s core defect lay in its multi-filament string. Unlike single-strand strings, this design allowed bacteria to wick into the uterus, leading to severe infections, pelvic inflammatory disease, infertility, ectopic pregnancies, spontaneous abortions, and tragically, several deaths. Thousands of women suffered complications, with over 2 million users potentially affected before the device’s withdrawal in 1974 amidst mounting evidence of its dangers.
The revelations sparked a torrent of lawsuits from injured women, exposing the manufacturer to immense legal liability. The sheer volume and severity of these claims ultimately forced A.H. Robins Company into bankruptcy in 1985. The scandal drew intense public and legislative scrutiny, galvanizing efforts to enhance oversight of medical devices, which had previously faced less stringent regulation than pharmaceuticals.
This crisis became a pivotal moment, directly leading to the Medical Device Amendments of 1976. These amendments significantly strengthened the U.S. Food and Drug Administration’s authority to regulate medical devices, requiring pre-market approval for high-risk devices, establishing performance standards, and mandating post-market surveillance. The Dalkon Shield scandal fundamentally changed medical regulations, raising ethical questions about corporate responsibility in healthcare.
The history of product safety is replete with cautionary tales, each scandal a grim reminder of the profound human cost and economic devastation that arise when corporate responsibility falters. From life-saving pharmaceuticals turning deadly to everyday vehicles becoming hazardous, and food products posing unseen threats, these incidents highlight a persistent challenge in safeguarding public health. They collectively underscore the indispensable role of vigilant regulatory oversight, ethical manufacturing practices, and transparent communication. Learning from these notorious chapters is not merely an academic exercise; it is a critical imperative that drives continuous improvement in product design, rigorous testing, and robust recall mechanisms, all aimed at rebuilding and maintaining the fundamental trust consumers place in the products they use every day. The ongoing evolution of consumer protection is a testament to the fact that while mistakes can be made, the commitment to safety must always remain paramount.



